Fascial Reconstruction Research
This project was introduced to the Bakken Medical Devices Center interns by Doctor Jaqueline Medina, she is specifically interested in the soft palate prosthetic rehabilitation.
Doctor Jaqueline Medina is a current clinical associate professor in the Department of Restorative Science. Dr. Medina’s clinical interests include ways to improve patient outcomes through digital technology and team approaches, utilizing digital technology in combination with conventional methodology.
With the guidance of Dr. Jaqueline Medina, a group of 4 interns: Seema Wadhwa, Helen Geng, Ali Al-Hinaai, Ishika Nimmagadda (left to right), researched and developed a medical device prototype to start the process to aid soft palate prosthetic rehabilitation.
Background Research
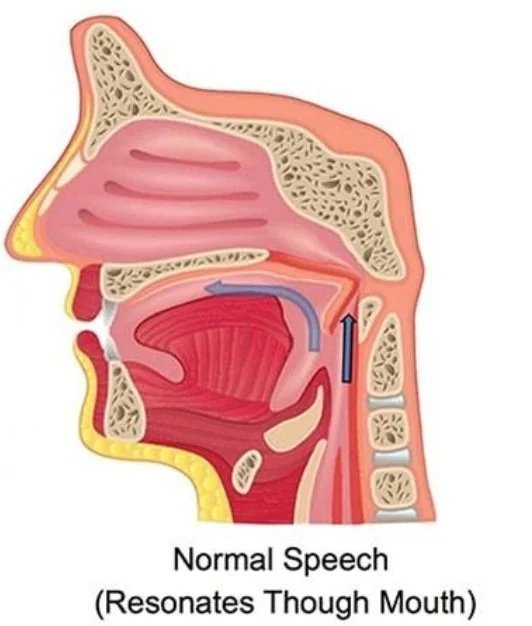
Oropharyngeal cancer is a disease that affects the tissues of the oropharynx (middle throat), treatable by removing the tissue affected through oral surgery. Most of the time, this leads the patient to lose their ability to to eat and speak normally. Velopharyngeal Insufficiency (VI) occurs when the soft palate does not close tightly to the back walls of the throat. When surgical correction is not an option, velopharyngeal obturators are used.
The obturator design highly depends on the remaining mouth anatomy of the patient. Conventional obturators are made out of acrylic resin and a cobalt alloy palatal framework, and the front is attached to a metal alloy. Existing obturators do help in tightly closing the soft palate, but they are heavy and rigid. This causes discomfort and the patient’s ability to speak and swallow normally may still be hindered.
Figure 1: Velopharyngeal Insufficiency
“Patients suffering from post-surgical tumor removal need a reliable way to restore their velopharyngeal defect that conforms to their existing anatomy in order to improve quality of life and to minimize reconstructive surgical intervention.”
~ Needs Statement
Ideation & Prototyping
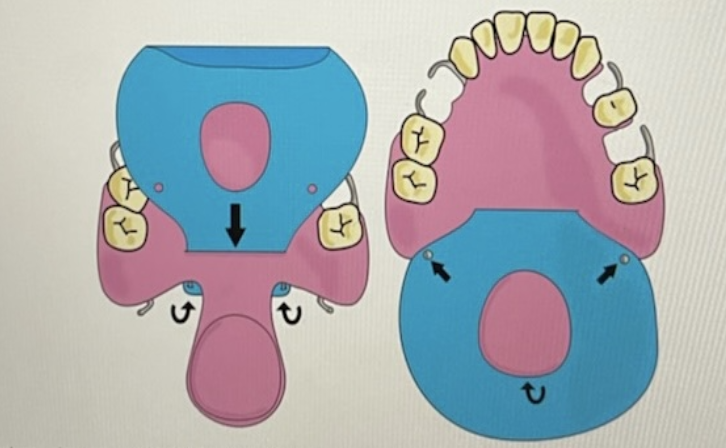
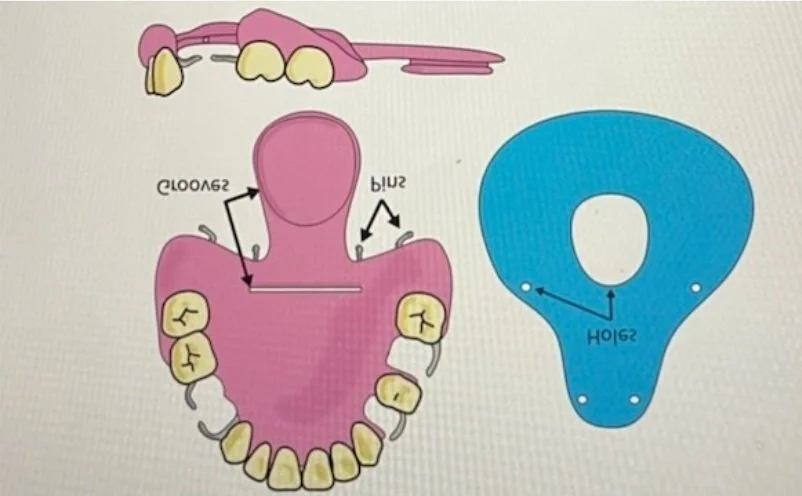
After conducting numerous interviews with patients and clinicians, through analyzing the data collected from the surveys, the team brainstormed five different ideas to improve on the design of current obturators. These were: 3D printed template, 3D printed patient specific, frame or mesh with support, expansion or inflation, and material filling or stacking. After preliminary sketching, discussion, and a design matrix, the team decided to design a 3D printed patient specific palatal obturator with three different sizes. A clinician can then choose the most suitable size for the patient.
Initially, clay was utilized to visualize the idea (Figure 2). For prototype 2, a 3D printed soft palate using PLA resin and LulzBot TAZ 6 3D printer was attached to a pre-made dental model (Figure 3). For prototype 3, a patient scan was segmented to create a 3D drawing of the patient defect (Figure 4). Then, the sketched defect was implemented to create an obturator.
The obturator was printed using Flexible 80A and Elastic 50A resins using a FormLabs printer (Figure 5). Upon further testing and inspection, the team decided to opt for Elastic 50A resin for the final product due to its malleability and lighter weight. To reduce the weight further, the bulb of the obturator was made hollow in prototype 4. A patient mouth cavity and head were segmented and 3D printed with Flexible 80A and BioMed clear resin for modeling and presentation purposes.
Figure 2: Clay Model
Figure 5 (right): Flexible 80 Obturator (right), Elastic 50A Obturator (left)
Figure 4 (above): CAD Model Figure 3 (top right): PLA Soft Palate
Competitive & Market Analysis
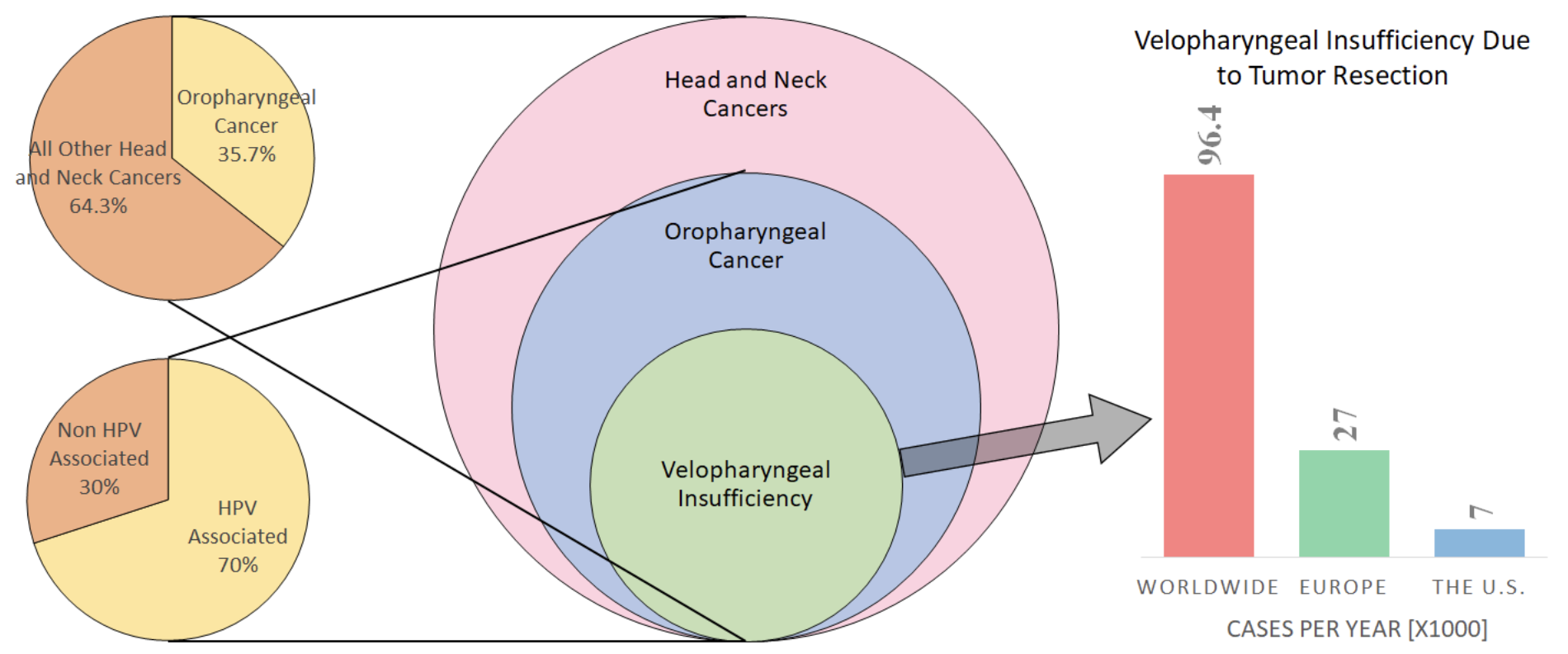
Oropharyngeal cancer patients who have had tumors surgically removed from the soft palate are the most common recipients of obturator prostheses, specifically those with cancers originating from human papillomavirus (HPV). An estimated 30% of patients with oropharyngeal cancer are also diagnosed with subsequent velopharyngeal insufficiency (VPI). For many who receive obturators in the event that other interventions are not feasible, the cost of $8,000 may pose as a financial burden as these procedures may not be covered by insurance companies.
Because anatomy and disease progression are different in every patient, treatments need to be specific to patients. This renders specific treatments as well as clinicians and healthcare providers as competitors, as opposed to medical device companies. The Suersen and Membranous Obturators Evaluation clinical trial appears to be the most direct competitor to our project, aiming to create a less rigid obturator.
Figure 6 : Seursen and Membranous Obturators Evaluation
Figure 7: Targeted Market
Next Steps
The next steps would be to conduct material testing to assess biocompatibility and to look into materials that are more optimal for the oral environment. Additionally, bench testing on tensile strength and interface retention forces are necessary to evaluate the stability and efficacy of the obturator. Collecting patient reviews and evaluations are also the next critical steps, which will determine any need for redesigning of the obturator.